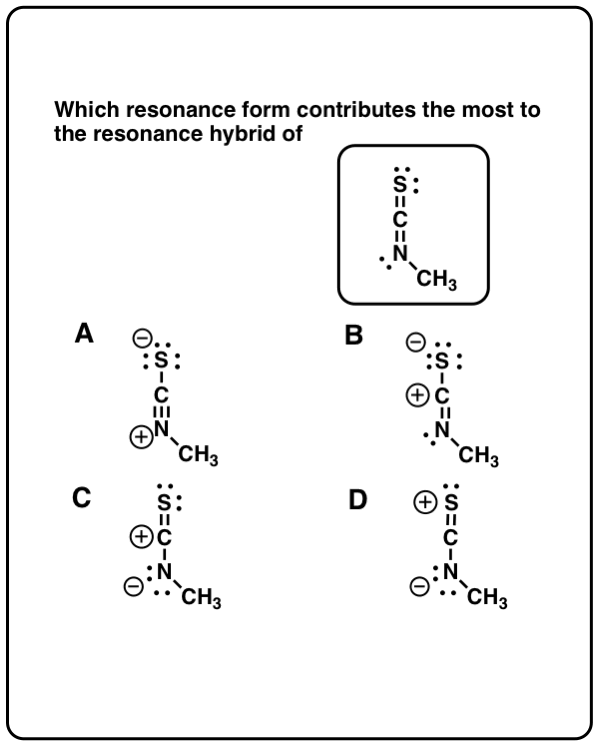
Calculate the formal charge in the “new” structure and label any non-zero formal charges.Ian Hunt, Department of Chemistry, University of Calgary. The “new” resonance structure should be a “product” automatically obtained by following the arrows. The resonance energy of a compound is a measure of the extra stability of. necessary to comprehend the meaning of curved arrows in drawing the resonance structures of organic. Use curved arrows to indicate the electron movement in the “original” resonance structure. organic chemistry particularly resonance concept is a hard.To move electrons, only π electrons and lone-pair electrons (NEVER move σ bonds!) can be moved from the higher electron density area to lower electron density area by following one of the three transformations:.(Formal charges on individual atom could be different, but net charge, that is the sum of all the charges, must be the same.) All resonance structures have the same number of electrons and net charge.All resonance structures must have the same atom connectivity, and only differ in the electron arrangement.(Keep in mind that all the rules applied to Lewis structures still apply here!) All resonance structures must be valid Lewis structures.Guidelines for Drawing Resonance Structures: EMR is very similar to the two other resonance techniques that take place here at the lab: nuclear magnetic resonance (NMR) and ion cyclotron resonance (ICR). EMR stands for electron magnetic resonance. Electrons, which are like tiny magnets, are the targets of EMR researchers. It is an analytical technique used to determine the purity and the composition of an element or a compound. As explained by EMR Facility Director Steve Hill. There are some very important rules we need to follow for such purposes. Nuclear Magnetic Resonance or NMR is a technique that makes use of magnetic fields and electromagnetic frequencies to study the molecular structure of a sample. Resonance : Resonance in organic chemistry refers to the organic compounds. Therefore, to predict whether the resonance effect applies or not, we usually need to construct “new” resonance structures (contributors) based on the “original” one that is available. Which of the following statements is best explained by resonance Group of. Here we will focus on how to draw resonance structures (or resonance contributors) for organic chemistry species, and how to compare the relative stabilities between the structures.Īccording to resonance effect, the greater the number of resonance contributors, the greater the resonance stabilization effect, and the more stable the species is. The overlap of two s orbitals (as in H2), the overlap of an s orbital and a p orbital (as in HCl), and the end-to-end overlap of. 
The discussion of resonance effect heavily relies on the understanding of resonance structures. Resonance stabilization effect (also known as resonance effect), as briefly mentioned in Section 1.3, is one of the fundamental concepts of Organic Chemistry and has broad applications.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
